What Are Natural Colors?
Natural colors are created or extracted from edible sources like fruits, vegetables, seeds, and minerals for the purpose of coloring food or beverage applications. However, there is no formal definition of “natural colors” by food authorities, like the Food and Drug Administration (FDA) in the United States or the European Food Safety Authority (EFSA) in Europe.
The Natural Food Colours Association (NATCOL) offers a similar definition saying they “originate from a wide range of sources like vegetables, fruits, plants, minerals and other edible natural sources.” They have been used for many years and are considered safe for use in food and beverage applications.
There are many different types, but check out some of the most used colors below, or read more about all the different types here.
Anthocyanins
from sources like:
purple corn, purple carrots, purple sweet potatoes, radishes, and elderberries
Betanins
from:
beets
Carbon Black
from:
charred vegetable matter like coconut husks
Carotenoids
from sources like:
annatto, beta-carotene, and paprika
Carmine
from:
the cochineal insect
Curcumin
from:
Turmeric root
Chlorophyll
from:
grass
Phycocyanins
from:
spirulina
Safflamins & Carthamins
from:
Safflower
Synthetic colors, on the other hand, include FD&C Red #40, Yellow #5, Blue #1, etc. (or Allura Red, Tartrazine, Brilliant Blue, etc.), and are created from non-edible sources, typically petroleum. While they are generally considered safe for food use, there have been studies linking them to hyperactive behavior in children and require special labelling in the EU.
Natural colors are made by removing the pigments from the natural sources through selective physical and/or chemical extraction methods. This means that the resulting material contains primarily pigments from the natural color source and excludes any flavors or nutritive elements.
The resulting colors are concentrated and standardized so food and beverage manufacturers receive the same color each time they place an order and can expect consistent results. See how it’s done below!
Want to try a sample? Request one here.
How is Caramel Color Made?
In simple terms, caramel color is made by cooking carbohydrates. It is similar to how you would make caramel on a stovetop – you heat sugar until the color changes from white to dark brown. But in order to create large quantities that are stable and suitable for coloring foods and beverages a few more steps are required. Watch the video below or read on for an in-depth look at how the most commonly used color in the world is made.
Need a quick overview? Check out the infographic!
1. Select your sugar
The first step in creating caramel color is to determine a carbohydrate source. There are many types of sugars that can be used to create caramel colors – sucrose, fructose, glucose, invert syrup – all derived from sources such as corn, wheat, sugar beets, and sugar cane. Some types of sugar are chosen because they work best for different classes of caramel, while other types may be chosen to meet certain certification requirements – such as non-GM or organic.
2. Determine your class of caramel
Next, the type of caramel is determined – there are four classes of caramel color, each with their own properties and requirements. Each class requires different reactants, or caramelization aides, such as food-grade acids, alkalis, and salts. Reactants help start the browning process, but also affect the stability, color intensity, and hue of the caramel color.
3. Cook the Sugar
Once the type of sugar and class of caramel is determined, the sugar is loaded into large cookers and a highly specific cooking process begins. For lower viscosity caramels, a system with high heat as well as pressure are used. These caramels are great for usability in manufacturing. For higher viscosity caramels, like class III caramels going into soy sauce, a non-pressurized system is used. While the sugars are heated, the reactants are added into the cookers at strategic times throughout the cook.
For the actual cooking process, caramel colors are created using either one or both of the following types of browning methods: 1) caramelization, or the browning of sugars, and 2) the Maillard reaction, the browning of sugars in the presence of amino acids or other ingredients with amine groups. While this may sound complex, the Maillard reaction is a normal browning process that occurs in the foods we enjoy every day – roasted coffee, seared steaks, and baked bread, for example.
Class I, or plain, caramel colors are created using solely caramelization to produce color. Class II, III, and IV caramel colors, on the other hand, use caramelization as well as Maillard browning reactions to achieve darker colors with increased stability. As the sugars brown, the reactants are cooked off and are not found in the final caramel color product.
After the sugar has finished cooking, the caramelized product is cooled and filtered to ensure product quality and uniformity. From here it can either be dried for use in powder form or moved onto packaging and shipping to customers.
Interested in learning more about caramel colors? Check out these resources:
How Much Do Natural Colors Cost?
The cost of natural colors varies, especially when compared to synthetics. Caramel colors, for example, are on the low end of the spectrum because they are made from raw materials (corn, wheat, sugar cane) that can be grown on a large scale, while spirulina is typically a high-cost color because it is grown in highly controlled environments and extracted using specialized processing equipment.
Other factors that influence the cost of natural colors include country of origin, agricultural variables, quality and other certifications, contracting ahead, and the need for special packaging. Even different forms of the same natural color can have different costs depending on how it’s made and where it’s made and how its packaged – making it impossible to provide a precise number.
Even though it isn’t possible to provide an exact number for any one color, we’ll talk about the main factors that influence the price of a natural color.
Country of Origin
Natural colors are grown all over the world and the cost of labor is different in each region. While lower labor costs may offer a cost advantage for your product, the standards employed may not meet certain sustainability or best practice requirements. So, you may opt to source from a more expensive region that has guarantees of fair trade and labor.
Geographical proximity of the raw material to the manufacturing site is another cost factor. If the distance between where a raw material is grown is far from where the color is extracted/manufactured, expect a significant increase in cost on to cover transportation costs, especially if it requires refrigerated or frozen shipping.
Certifications / certificates
Organic, Kosher, Halal, and Non-GM are all great certifications for products to have. But they require extra costs related to manufacturing practices and certifications by organizations to ensure that the products meet certain requirements.
Agricultural Variables
Weather
Just like other agricultural products, the price of produce grown for natural colors is subject to the weather – creating abundant yields or measly harvests. Rain can be beneficial to crop size and quality, increasing yields and lowering the cost. But too much rain can flood fields and damage plants, increasing the price.
Drought can also affect the yield, but almost always negatively. Even short-term drought during the key stages of growing can stunt growth and result in poor size and quality of produce. When this happens, the price of the natural color will increase because it will almost certainly lead to a shortage.
Similarly, climate change is affecting crops all around the world. While some plants may benefit from warmer weather or more carbon dioxide, most crop growing regions will struggle with consistently warmer weather, longer more frequent droughts, and an increase in severe weather.
Pests and disease
Insects and wildlife, such as rats, mice, and birds can all threaten the health of growing crops. Insects can damage the leaves, roots, and stems of growing plants, affecting their ability to produce fruits and vegetable. The elderberry crop in 2020 is a prime example of this – the crops were completely devasted by a type of fruit fly.
Wildlife can damage fruits or vegetables before and even after harvest in barns or storage containers. The most affected crops are wheat and corn, the main sources used to produce caramel colors and burnt sugars.
Bacterial, fungal, and viral infections can also cause significant damage to crops. The symptoms of disease, such as leaf wilt or damage, lesions, and spots, can negatively impact a plant’s ability to grow and produce healthy fruit/vegetables and requiring measures to prevent the spread – all of which increase the cost of the raw materials.
Ability to Contract
Because the biggest factor in the cost of natural colors is related to growing crops, a little planning goes a long way. If you are able/willing to contract for your annual needs, this gives the supplier time to source and produce to scale in the most economical way – and pass the savings on to you in the process.
Order/Pack Sizes
Order size can also help reduce cost. Larger orders allow for overhead costs related to production to be spread over more material, resulting in a lower cost per pound. Smaller orders, on the other hand require the same production cost to run the machines, but because there is less material, the cost per pound will be higher. It’s usually more expensive to “spot buy”-if not in price, in quality.
Pack size and type can also make a difference in your final costs. It requires longer equipment runs and more labor to fill 4–gallon jugs than a single large tote, for example. And specialized films and wraps add additional labor and material costs to order.
Despite all the influences on the cost of a natural color, your supplier should be able to work with you to navigate different options to get the best order size and packaging to fit your budget. Contact us to get started.
Is Caramel Color Safe?
Is caramel color safe?
Is caramel color safe? The answer is yes – caramel colors have been deemed safe by all major global food regulatory bodies, including the US Food and Drug Administration (FDA), the Joint FAO/WHO Expert Committee for Food Additives (JECFA), Codex Alimentarius, the European Food Safety Authority (EFSA) and Health Canada. Here’s why:
Background
The question around the safety of caramel color arose when California added a chemical found in class III and IV caramel colors called 4-Methylimidizole, or 4-MEI, to their Prop-65 list. Adding it was based on a controversial study by the National Toxicology Program (NTP) from 2007. The 2-year study on mice showed an increased incidence of certain lung tumors when they consumed 4-MEI. However, the levels of 4-MEI given to the mice far exceeded the normal amount humans would be exposed to when consuming food or beverages – an amount equivalent to a human drinking thousands of cans of cola every single day throughout their life.
What is 4-MEI?
4-MEI is a chemical compound that naturally forms during the cooking of food and beverage items we consume on a regular basis: coffee, cooked meat, baked goods, etc. Since caramel colors are created by cooking sugars, 4-MEI is naturally formed during the manufacture of certain products – specifically class III and IV caramels. 4-MEI is not present in Class I and II caramels and 4-MEI itself is never added to foods or beverages.
4-MEI occurs in very low levels in caramel colors. And since caramel is often used at dosage rates of around 0.1%-2.0% in final products, the occurrence of 4-MEI from caramel color in finished goods is miniscule.
But since it was added to the Prop-65 list, in order to comply with California law, many caramel manufacturers (including us!) began to innovate new methods of cooking class III and IV caramel colors that resulted in even lower levels than already occur, called ‘low 4-MEI’ caramels, some of which have levels so low they can be difficult to detect in the caramel color, let alone the finished product.
Safety Studies
In order to ensure continued consumer safety, many studies on the safety of caramel color and the recommended daily intake levels on 4-MEI have been carried out since the controversial 2007 NTP study was published.
In a review of the scientific literature on 4-MEI, EFSA found that the highest exposure level to 4-MeI that could result from the consumption of foods containing class III and IV caramels “was not concerning.”
Interested in reading more about the safety of caramel color? Check out these resources:
What are E-Numbers (E#s)
We often get questions from our customers such as ‘What are E-numbers?’ ‘Are E-numbers bad?’ ‘Why do some natural colors have E#s while others don’t?’ There is even a common misconception that an E# is another term for an artificial food additive, but this is far from the truth. Don’t worry – E#s are nothing to fear, and this is why:
The Origin of E#s
E–Numbers or E#s – short for ‘Europe Numbers’ – are simply a unified list of food additives and ingredients that was first compiled in the early 1960s. This list includes categories for colors, antioxidants, preservatives, and other common food ingredients. E-numbers were designed to show that an additive or ingredient has been tested as safe and approved for use by the European Food Safety Authority (EFSA) within the European Union. But you can also find E-Numbers on food labels from many countries outside the EU, including Australia, the Gulf States, New Zealand, Russia, and South Africa.*
Which Natural Colors have E#s?
This list includes all the natural colors that are approved for use in the EU – including carotene from carrots, lutein from marigold petals, and anthocyanins from a variety of sources, like elderberry and purple carrot. In order to be included in this list of approved food additives and to qualify for an E#, these ingredients must first pass rigorous safety studies and an approval processes through EFSA that often takes years!
E# Use on Labels
When used on a food label, an E# gives the final consumer access to information on what is in their food and the confidence to know that it has been tested and is safe for consumption. However, ingredients that have an E# are not limited to being labeled as just the E#. Manufacturers also have the option to label the full name of the ingredient. ‘Colours: copper chlorophyll’, for example, instead of ‘colours: E 141.’
*While these countries technically reference ‘INS numbers’ used by Codex, many INS and E numbers correspond because EU and Codex are closely aligned.
What is Caramel Color?
Caramel Color: What is it?
Caramel color is the most widely used non-synthetic color in the food and beverage industry. It is created through the controlled heating of sugars from sources such as corn, wheat, and sugar beet and they can range in hue from golden to dark brown.
why are there four classes?
Approved food-grade reactants, such as alkalis, acids, or salts, are used to promote caramelization. Based on the type of reactant that is used, the caramel is divided into one of four different types, or classes. They are: Class I (E150a), Class II (E150b), Class III (E150c), and Class IV (E150d). Each class of caramel color has different specifications on color intensity and hue. And while they are all favored for certain application, caramel color generally has excellent heat, light, and acid stability and works in many different applications.
Class I Caramel Color, E150a
Class I caramel colors, also known as plain caramels, are created by cooking a carbohydrate, typically glucose or sucrose, with acids, bases, or salts – such as citric acid. They generally range in hue from yellow to red-brown and have a slight negative colloidal charge.
While most class I caramel colors are only stable above pH 3.0, some are stable down to pH 2.8. These options do not contain 4-MeI or sulfites and have the highest stability in alcohol compared to the other three classes. They can be used in most applications: from baking to confections to beverages. Recently, demand for class I caramel colors has increased due to consumers looking for products with simpler labels. That’s why innovations in class I’s have focused on creating colors that can reach similar shades or use rates to Class III and IV caramels. Learn more about these advancements here.
Class II Caramel Color, E150b
Class II caramel colors, or spirit caramels, are created by cooking a carbohydrate with the aid of food grade sulfites. The hues can range from very yellow to dark red-brown and most are stable above pH 3.0. Class II caramels have a negative colloidal charge.
Like class I caramels, this class does not contain 4-MeI. They exhibit good stability in alcohol and are most commonly used in cognac, sherry, and brandy, giving them the name “spirit caramels”. Despite their excellent alcohol stability, these caramels are not frequently used because similar results can be obtained with class I’s.
Class III Caramel Color, E150c
Class III caramel colors, also called ‘beer caramels’, are created through the controlled heating of carbohydrate sources with food grade ammonium compounds. The resulting color ranges from a light brown to dark red-brown.
Class III Caramel Colors do not contain sulfites and have a positive colloidal charge in most food applications. They are typically beer stable and salt stable, making them a favorite for brewers looking to standardize dark beers or soy sauce and bouillon manufacturers that require a color that will be stable in a 20% salt solution.
Class IV Caramel Color, E150d
Class IV caramel color is the most widely used and versatile of the caramel colors. It is created by cooking a carbohydrate with food grade ammonium and sulfite compounds. The color ranges from a light brown to deep black-browns. Class IV caramels exhibit a strong negative charge over a wide pH range, making them the most versatile caramel colors. They are widely used in the soft drink industry due to their low dosage requirements and acid stability. We offer a wide variety of class IV options, including single-strength, double-strength, and low 4-MEI. Because this class of color contains 4-MEI, some organizations have called into question the safety of caramel color. Learn more about the safety of caramel color and new innovations here.
Ready to try it? Request a sample here.
Interested in learning more about caramel colors? Check out these resources:
The Types of Natural Colors
The demand for natural colors has increased as consumers look for simpler, more understandable product labels. And while these can come from a wide variety of plant and mineral sources, there are several main types of natural colors that are most commonly used in foods and beverages. Below we’ll take you through the sources you are most likely to find in foods and beverages and how they perform in application.
Anthocyanins

Anthocyanins are water soluble natural pigments responsible for the pink, red, purple, and blue hues of many flowers and edible fruits. The anthocyanin sources most commonly used for natural color production are purple corn, purple carrots, red radish, and elderberry. Other sources like cabbage and purple sweet potato can also be used.
Within each plant, these pigment compounds vary in concentration and exact chemical structure making them all slightly different when using them for color in a food or beverage. For example, purple sweet potato appears bright magenta pink in low pH applications, while others like purple corn appear bright red. They are all generally heat and light stable but anthocyanins from vegetable sources tend to be slightly more stable than those from fruit sources.
Anthocyanins are unique compared to other natural colors because the molecule slightly shifts in structure based on the pH that it is in, causing it to change colors from pink/red in lower pH levels to purple/blue in neutral/high pH levels. Watch it in action!
Betanins

Betanins are the water–soluble pigments that give red beets, or beetroot, their bright red hue. They provide magenta pink to red shades in foods and beverages and are one of the most popular natural color sources, especially for applications like confections, yogurt, and frostings. These colors are generally very light stable but will fade during heat processing.
Red beet is often used as an alternative to Red #40 or Carmine when product developers need a vegetarian or Kosher friendly red source, but, because it can fade during heat processing, it is not ideal for applications like cakes and other baked goods that are subjected to high heat. You can learn more about the best alternatives to carmine for different applications here.
Carmine & cochineal

Cochineal and carmine are natural colors extracted from the cactus-dwelling female cochineal insect that is native to Latin America. Depending on the method of extraction, it can range in hue from orange to red to purple.
In terms of stability, this natural color source is one of the best – it has excellent heat, light, and pH stability and works well in a wide range of food and beverage applications, making it popular as a Red #40 replacer when kosher certification is not a requirement.
Carotenoids

Carotenoids are one of the most frequently used types of natural colors. They are the pigments responsible for giving plants their red, orange, and yellow shades. For food and beverage purposes, they come from a wide range of natural color sources, including annatto, beta-carotene, and paprika.
Like Anthocyanins, within each plant, these pigments vary in concentration and exact chemical structure making them all slightly different, but they are widely used in the food industry because they are generally stable to heat, light, and pH. They are most commonly used in beverage, dairy, and savory applications or as natural alternatives to Yellow #5 and #6.
Chlorophyll

Chlorophyll is naturally found in the chloroplasts in green plants and its abundance in nature and vibrant green shade make it ideal for use as a natural food color. When originally extracted it is in an oil soluble form called chlorophyll but can be converted into a water–soluble version called chlorophyllin. Since chlorophyll[in]s are not the most stable or vibrant options for food or beverage applications, they can be stabilized by ‘coppering’ – creating copper chlorophyll and copper chlorophyllin. Learn more about the differences between these pigments here.
These are vibrant green options that have good stability to heat, light, and pH.
Curcumin

Curcumin, also called turmeric, is a vibrant yellow pigment extracted from the root of the turmeric plant, which is in the ginger family. Turmeric has a long history of use as a spice and as a medicinal supplement.
The extracted color has excellent heat stability, making it perfect for applications like bakery, soups, seasonings, and extruded snacks as well as frostings and confections. However, it performs best when it has opaque packaging because it will fade when exposed to light. Because this pigment is so vibrant, it can be used to create close matches to Yellow #5.
Phycocyanins

Phycocyanins are one of the newest types of natural colors to be approved but are already widely used in the food industry. They are the water-soluble pigments responsible for the blue color extracted from the algae Spirulina. This algae occurs naturally in freshwater and marine habitats and has a long history as a food supplement in many countries.
The extracted color is a vivid blue without any green or purple undertones. Despite its bright shade, Spirulina has limited uses because it is susceptible to fading in heat and light and is only stable between pH 4-7. It is most successfully used as a natural alternative to Blue #1 in applications such as ice creams, confections, and dry-mix beverages.
Why Do Anthocyanins
Change Color
What are anthocyanins?
Before we dive into why anthocyanins change color, it’s important to know what they are. Anthocyanins are natural, water soluble pigments responsible for the blue, purple, pink, and red colors in many fruit and vegetable sources. In the food coloring industry, they are typically extracted from sources such as purple corn, purple carrots, radishes, elderberries, and other fruits and vegetables that are bred specifically for their high pigment concentrations.
When used in an application with a lower pH, like confections or beverages, anthocyanins will appear bright red to pink. But when that same anthocyanin is put in an application that has a higher pH, such as a cupcake or its frosting, the anthocyanin will appear bluish-purple. But what is causing this color shift? Why can’t the anthocyanin color work the same in every application?
It all has to do with the natural reaction the molecule has to the pH of its surrounding environment.
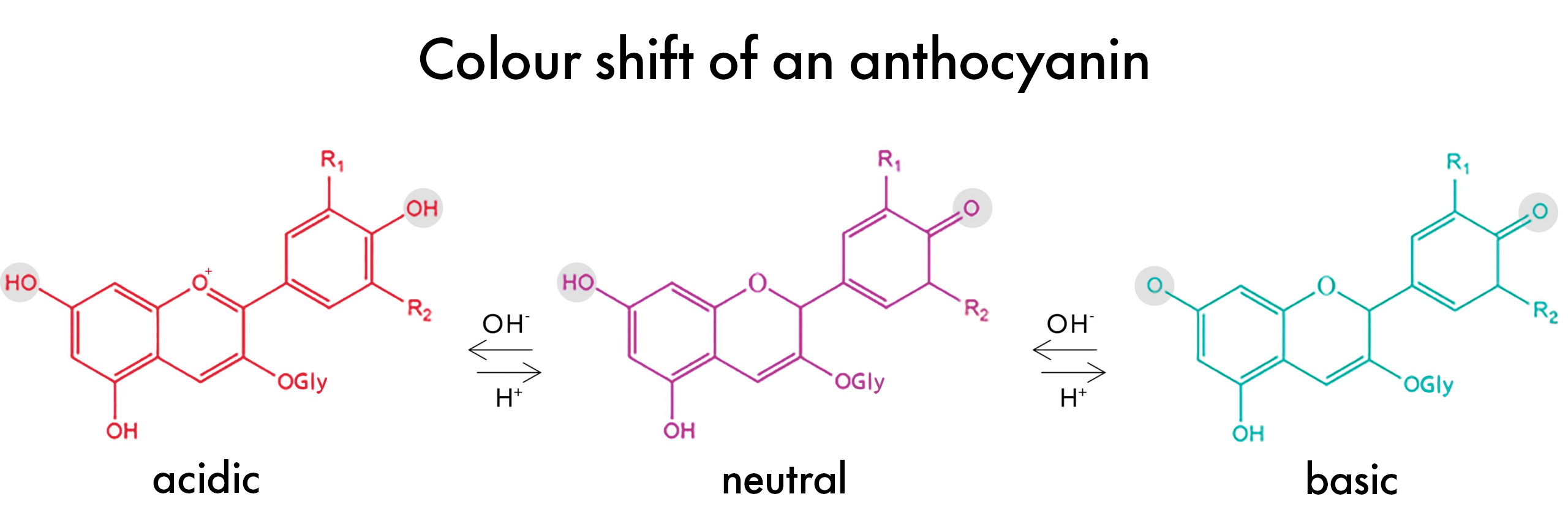
So why do Anthocyanins Change Colors?
Anthocyanins change color in different pH levels because their molecular structure actually shifts as the pH of the solution they are in changes from acidic to basic and vice versa. This makes these pigments unique compared to other natural colors.
At a low pH of around 3, the anthocyanin molecule is ‘protonated’. This means the phenolic -OH groups (highlighted grey in the figure above) have hydrogens. In this environment, the anthocyanin is a positive ion, or cation. It absorbs light in the blue-green spectrum (approx. 450-560nm) and appears red to the human eye.
As the pH of the environment increases, however, the anthocyanin molecules become ‘deprotonated’ –protons are removed from the phenol groups – and the light absorption of the molecule shifts, now absorbing light in the yellow-orange spectrum (approx. 570-620nm) giving it a blue-ish purple appearance to the human eye.
Decrease the pH again, and the protons re-bond to the molecule therefore changing the light absorbance once again and reverting the color back to the original red to pink. And that’s how Anthocyanins change color…Pretty cool, right?
Want to get started with a sample? Click here.
How Are Natural Colors Made?
How are natural colors made?
Just like how natural colors come from a wide variety of places, they can also be made in a number of different ways. A lot of how a natural color is manufactured is based on whether the pigment is oil soluble or water soluble.
The different solubilities require different methods for removing the color from the natural source. Extraction methods can also vary based on how the color is contained within the source – whether the color is on the exterior part of the crop, in the flesh, or part of the seed. We’ll talk about the different types of extraction below. After extraction, the colors are concentrated for maximum vibrancy with low use rates.
Water Soluble Natural Colors
Juicing, Griding, & Crushing
When the color is water soluble and is contained within the fruit of the source – like with red beets or purple carrots – physical methods, such as juicing by crushing or grinding, are most commonly used. In this processing, the beets or carrots are run through a machine that crushes them until all the juice is extracted. The juice can then be concentrated down to different levels and either kept in liquid form or sent on to a dryer to make powders.
Water Extraction
When the color lies on the outside of the natural source, like with purple corn, carmine, and spirulina, water extraction is a great option. In this process the natural source is soaked in water. Since the pigment is water soluble, it easily dissolves, creating a colorful liquid that can then be concentrated to different levels using an evaporator. Similar to the grinding or crushing method, these colors can then be sent on for drying or be kept in liquid form.
You can see this method in action by watching our video on how we make our Amaize® Red.
Oil Soluble Natural Colors
Solvent Extraction
Oil soluble pigments, such as beta carotene, annatto, paprika, and turmeric, are typically found within the plant/organism, and often require what is called ‘solvent extraction’ in addition to crushing or grinding.
The first step is to crush or grind the source. A solvent is then used to separate the pigment from the plant because the pigment isn’t able to dissolve in water. While the word solvent may sound scary – don’t worry – the solvent can be as simple as vegetable oil or alcohol which can be used to dissolve oil-based pigments (water is the ‘solvent’ in water extraction). These can also include several stronger organic solvents that are approved for use in food.
Like the water soluble natural colors, once the color has been obtained, the solvent is evaporated out, and the color is diluted to different color strengths.
Whichever type of method is used, it is important to extract and concentrate the color as soon as the pigment has been disturbed (such as by crushing) in order to maintain peak quality and vibrancy.
